This is opposed to static equilibrium where there is no reaction at all. When you put salt in water particles leave its surface and it goes into solution dissolves.

Dynamic Equilibrium 3 3 2 Edexcel Igcse Chemistry Revision Notes 2019 Save My Exams
Presley will be equal to the rate off.

. J IQ e ee 3. The exact moment of equilibrium happens when the rate of the forward reaction equals the rate of the reverse reaction. Why does a larg Keq value mean that the products are favored in an equilibri large no rruxar pro sys em.
Cecacú0-Ys and pr080cts are conhnuoosl bein fDrmzd rakðe 2. A dynamic equilibrium is established in a solution when particles return to the surface of the solid at the same rate as they are leaving. This was already explored in the prior learning objective in the example involving compounds A B C and D.
Dynamic state of equilibrium is a more accurate description where internally things may fluctuate but will always stay in specific limits. So in this question um they asked us why the world dynamics used to describe a nickel Librium in order to understand that lets take a look for example the reaction that if we have a plus B and in equilibrium with C plus D so at it will librium the rate off. Static equilibrium is an equilibrium that occurs when all particles in the reaction are at rest and there is no motion between reactants and products.
1aDescribe the dynamic equilibrium that exists in the twowater tanks. B Dynamic equilibrium is called dynamic because the forward and reverse reactions are still occurring however they are occurring at the same rate. A Dynamic equilibrium is called dynamic because the forward and reverse reactions are still occurring however they are occurring at the different rate.
It means the rate of forwarding and the backward reaction is equal to each other. In this case the object will remain in motion with constant velocity. It is dynamic because there are many factors that affect what that ratio will be as defined by LeChatelier.
A forward reaction which is a plus B from C. The stability attained is actually a dynamic equilibrium in which continuous change occurs yet relatively uniform. Chemistry questions and answers.
However you can add so much salt that it will not all dissolve. C Dynamic equilibrium describes an object in motion having equal and balanced forces acting upon it- An object is said to be in dynamic equilibrium if it is in motion and all the forces acting on the object are balanced so that the net force resultant of the forces acting on the object is zero. Synonyms for Dynamic equilibrium.
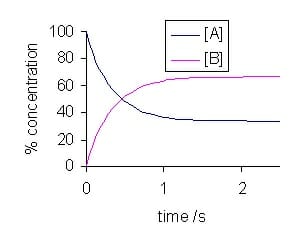
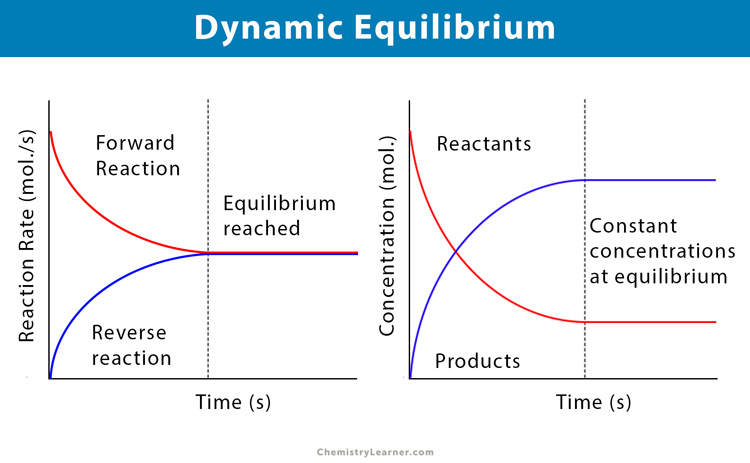
Given the fact that the concentrations of reactants and products are not changing why is the word dynamic used to describe chemical equilibrium. The word dynamic is used to describe chemical equilibrium because eventhough the concentration of reactants and products remains constant the reaction does not stop but still continues with the rate of forward reaction equal to the rate of backward reaction. These equations are dynamic because the forward and reverse reactions are still occurring but the two rates are equal and unchanging so theyre also at equilibrium.
And this equilibrium does not need to occur right in the middle of two floorsyou could be near the bottom near the top or anywhere in between when you carry out your reverse process. Van t Hoff on page 162 in his classic 1884 Études de dynamique Chimique used the phrase principle of mobile equilibrium to describe what we now use dynamic for. BExplain how LeChateliers principle applies when thefaucet on the right tank is opened.
Dynamic equilibrium is an equilibrium where reactants are converted to products and products are converted to reactants at an equal and constant rate. Like dynamic equilibrium static equilibrium is another type of equilibrium. Equilibrium is dynamicboth forward and reverse reactions continue even though the reaction appears to have stopped.
Does the following equation represent a homogene us equili ium or hetero eneous equilibrium. Why is the word dynamic used to describe a chemical equilibrium. Homeostasis any self-regulating process by which biological systems tend to maintain stability while adjusting to conditions that are optimal for survival.
Chemical equilibrium refers to the balance between products and reactants after a given reaction has reached a state of order in which both reactants and products are forming at a constant rate. Synonyms for Dynamic Equilibrium other words and phrases for Dynamic Equilibrium. Civil engineers use dynamic equilibrium during the construction of bridges or buildings.
Why is it called dynamic. At chemical equilibrium both forward and reverse reactions continue but because their rates are equal no net change occurs occurs in the concentration of the reaction components. Word dynamic used to describe ch mic I equilibrium.
If unsuccessful disaster or death ensues. The human being is a very complex organism in which the maintenance of life depends upon various parts. Dynamic equilibrium is an equilibrium where reactants are converted into products and vice versa at a constant rate constant rate of change.
CExplain how LeChateliers principle applies whenwater is added to the right tank. 32 other terms for dynamic equilibrium- words and phrases with similar meaning. Problem 35 Explain how a person bailing out a row boat with.
This is the meaning of the word dynamic in the title. Dynamic equilibrium only occurs in reversible reactions and its when the rate of the forward reaction is equal to the rate of the reverse reaction. If homeostasis is successful life continues.
The formation of the chemical complexes shows a dynamic equilibrium and the concentrations are rules by the stability constants of complexes.
Difference Between Static And Dynamic Equilibrium Biology Dictionary
Dynamic Equilibrium Definition And Examples

Dynamic Equilibrium 3 3 2 Edexcel Igcse Chemistry Revision Notes 2019 Save My Exams
0 Comments